 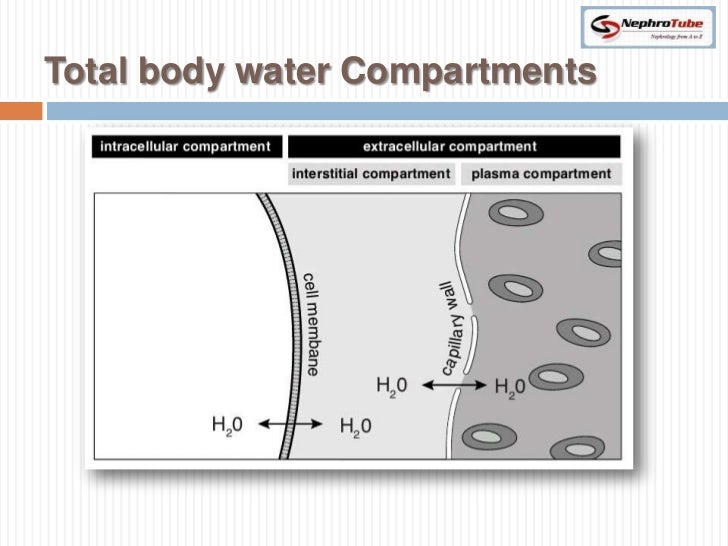
In the figure below, assume that all solutes contributing to the concentrations are nonpenetrating and that the cell membrane is permeable to water. Net water movement stops when the two compartments reach osmotic equilibrium. (By convention, we assume that all intracellular solutes are nonpenetrating.) Water will move into the compartment (cell or solution) with the higher concentration of nonpenetrating solutes. To determine the tonicity of a solution, compare the concentration of nonpenetrating solutes in the solution to the total concentration inside the cell. Tonicity of a solution is a term that describes how a solution will affect cell volume if that cell were placed in that solution. Solutes that can cross a cell membrane are said to be penetrating solutes, whereas solutes that cannot cross a cell membrane are said to be nonpenetrating. To predict if water will move across a cell membrane, you need to know whether the solutes in the solution can cross that membrane. Although osmolarity tells you the concentration of solute particles in solution, osmolarity cannot always tell you the direction of water movement across a membrane. Using the same beakers as Part E, how would you describe the osmolarity of beaker B relative to beaker C? Beaker B is hyperosmotic to beaker C. How would you describe the osmolarity of beaker A relative to beaker B? Beaker A is hyposmotic to beaker B. Same- Add 0.5 mosmol glucose and 0.5L water- Add 1 mosmol NaCl and 1 L water You have three separate beakers, each containing the following osmolarities: Increase- Remove 0.5 L of water- Add 1 mosmol of KClĭecrease- Add 1 L of pure water- Add 0.5 mosmol NaCl and 1 L water You must first consider the sum of the solutes, then consider the total volume before expressing the value as the concentration (solute amount/total volume) of the mixed solution.ĭrag the solutions to the correct box to indicate whether the final concentration of the mixed solution has increased, decreased, or remained the same. Remember that you cannot just add concentrations. Use the equation solute/volume = concentration (S/V = concentration) for each item to find the concentration of the added solution. Add or subtract the solutions or solute listed below, then determine the concentration of the mixed solution. 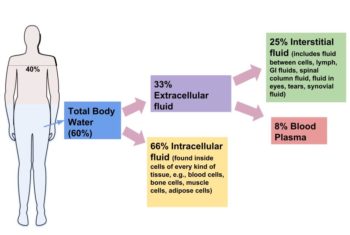 
Submit You have a beaker containing 1 L of 1 mOsM glucose. Presuming that the salts listed below completely dissociate in water, which of the following solutions below has the highest osmolarity? 7 mM sodium chloride (NaCl) In the human body, we express this concentration in milliosmoles per liter (mOsM). Using the same principles, one mole of NaCl dissolved in a liter of water would become a 1 molar (1 M) NaCl solution, but would also be a 1.8 osmolar (1.8 OsM) solution. Some salts, like NaCl, will incompletely dissociate into their respective ions when dissolved in water. Thus, osmolarity is the measure of the number of solutes per volume of solution. One mole of AB in water would be one molar (1 M), but it would be 2 osmolar (2 OsM) solution, because there would be 1 mole of each ion dissolved in that solution. For example, suppose a hypothetical salt, AB, completely dissociates into A+ and B- in water. Osmolarity is a way of expressing solute concentration taking into account whether or not the solute in question dissociates into ions when dissolved in water. Which of the following statements correctly describes the relationship between intracellular fluid (ICF) and extracellular fluid (ECF)? The types of solutes and their distribution between the ICF and ECF are not the same. Water moves down its own gradient into the compartment with the higher osmolarity until both compartments have the same solute-to-volume ratio (or osmolarity). If the osmolarity in one compartment changes, water moves by osmosis to bring the two compartments back to osmotic equilibrium. Osmotic equilibrium occurs because water passes freely through cell membranes using open channels or the water channels called aquaporins. This means that the ICF and the ECF have the same total solute concentrations, despite the differences in their specific solute composition. Neither- The fluid compartments of the human body are in osmotic equilibrium. Intracellular Fluid- cytosol of a red blood cell- cytoplasm of a neuronĮxtracellular Fluid- blood plasma- interstitial fluid Identify whether each of the following is a component of the intracellular fluid or extracellular fluid by dragging the box into the appropriate compartment. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
